Next-Generation Flow Battery Design Sets Records
A common food and medicine additive has shown it can boost the capacity and longevity of a next-generation flow battery design in a record-setting experiment.
A research team from the Department of Energy’s Pacific Northwest National Laboratory reports that the flow battery, a design optimized for electrical grid energy storage, maintained its capacity to store and release energy for more than a year of continuous charge and discharge.
The study, just published in the journal Joule, details the first use of a dissolved simple sugar called β-cyclodextrin, a derivative of starch, to boost battery longevity and capacity. In a series of experiments, the scientists optimized the ratio of chemicals in the system until it achieved 60 percent more peak power. Then they cycled the battery over and over for more than a year, only stopping the experiment when the plastic tubing failed. During all that time, the flow battery barely lost any of its activity to recharge. This is the first laboratory-scale flow battery experiment to report more than a year of continuous use with minimal loss of capacity.
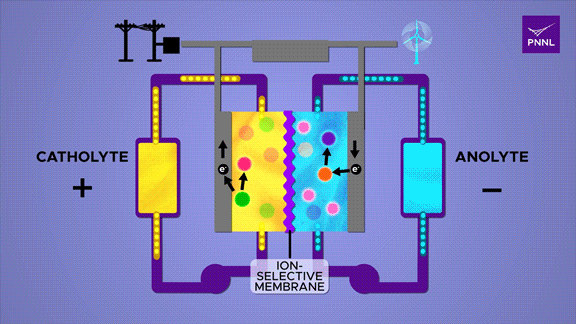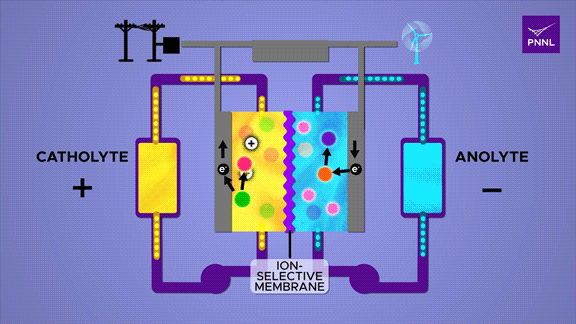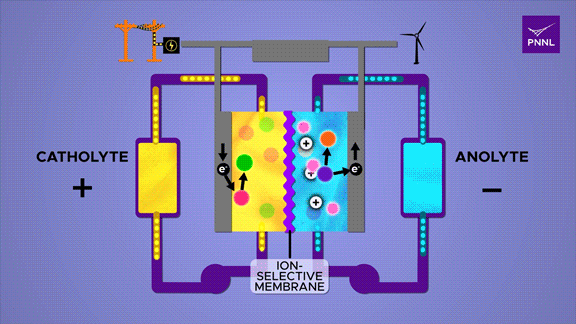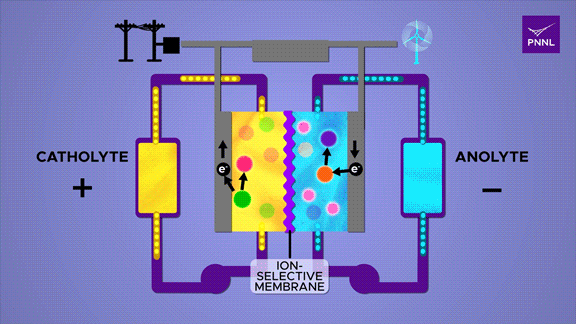
Flow batteries provide long-lasting, rechargeable energy storage, particularly for grid reliability. Unlike solid-state batteries, flow batteries store energy in liquid electrolyte, shown here in yellow and blue. Researchers at PNNL developed a cheap and effective new flow battery that uses a simple sugar derivative called β-cyclodextrin (pink) to speed up the chemical reaction that converts energy stored in chemical bonds (purple to orange), releasing energy (electrons) to power an external circuit. A parallel reversible process (red-green) in the positive catholyte solution balances the positive and negative charges during charge and discharge. (Animation by Sara Levine | Pacific Northwest National Laboratory)
The β-cyclodextrin additive is also the first to speed the electrochemical reaction that stores and then releases the flow battery energy, in a process called homogeneous catalysis. This means the sugar does its work while dissolved in solution, rather than as a solid applied to a surface.
“This is a brand new approach to developing flow battery electrolyte,” said Wei Wang, a long-time PNNL battery researcher and the principal investigator of the study. “We showed that you can use a totally different type of catalyst designed to accelerate the energy conversion. And further, because it is dissolved in the liquid electrolyte it eliminates the possibility of a solid dislodging and fouling the system.”
A benign ‘sugar water’ sweetens the pot for an effective flow battery
The PNNL research team that developed this new battery design includes researchers with backgrounds in organic and chemical synthesis. These skills came in handy when the team chose to work with materials that had not been used for battery research, but which are already produced for other industrial uses.
“We were looking for a simple way to dissolve more fluorenol in our water-based electrolyte,” said Ruozhu Feng, the first author of the new study. “The β-cyclodextrin helped do that, modestly, but it’s real benefit was this surprising catalytic ability.”

The researchers then worked with co-author Sharon Hammes-Schiffer of Yale University, a leading authority on the chemical reaction underlying the catalytic boost, to explain how it works.
As described in the research study, the sugar additive accepts positively charged protons, which helps balance out the movement of negative electrons as the battery discharges. The details are a bit more complicated, but it’s like the sugar sweetens the pot to allow the other chemicals to complete their chemical dance.
The study is the next generation of a PNNL-patented flow battery design first described in the journal Science in 2021. There, the researchers showed that another common chemical, called fluorenone, is an effective flow battery component. But that initial breakthrough needed improvement because the process was slow compared with commercialized flow battery technology. This new advance makes the battery design a candidate for scale up, the researchers say.
At the same time, the research team is working to further improve the system by experimenting with other compounds that are similar to β-cyclodextrin but smaller. Like honey, β-cyclodextrin addition also makes the liquid thicker, which is less than ideal for a flowing system. Nonetheless, the researchers found its benefits outweighed its drawbacks.
Understanding the complex chemistry happening inside the new flow battery design required the expertise of many scientists, including Ying Chen, Xin Zhang, Peiyuan Gao, Ping Chen, Sebastian Mergelsberg, Lirong Zhong, Aaron Hollas, Yangang Lian, Vijayakumar Murugesan, Qian Huang, Eric Walter and Yuyan Shao of PNNL, and Benjamin J. G. Rousseau and Hammes-Schiffer of Yale, in addition to Feng and Wang.
PNNL | https://www.pnnl.gov/









